1 min read
AGC Biologics to Expand Biopharmaceutical CDMO Capacity
Nick McDonald November 30, 2020 at 3:10 PM
Doubling the production capacity at its site in Denmark to address strong demand
(SEATTLE), November 30, 2020 -- AGC Biologics announced that it will increase the production capacity of its Copenhagen facility. AGC Biologics will construct a new building on land adjacent to its current Copenhagen site, adding production floors with 2000L single-use bioreactors, labs and office space. The total investment for this expansion is estimated to be approximately €160M, and the start of operation is scheduled for 2023.
The biopharmaceutical CDMO market continues to grow at around 10% annually, and the accumulation of contracts awarded to AGC Biologics is outpacing the market. This expansion will more than double AGC Biologics’ single-use bioreactor mammalian cell-culture capacity in Copenhagen, enabling AGC Biologics to meet the needs of the market. The new building will have a total floor area of approximately 19,000m2 and is an important part of AGC Biologics’ global expansion plan.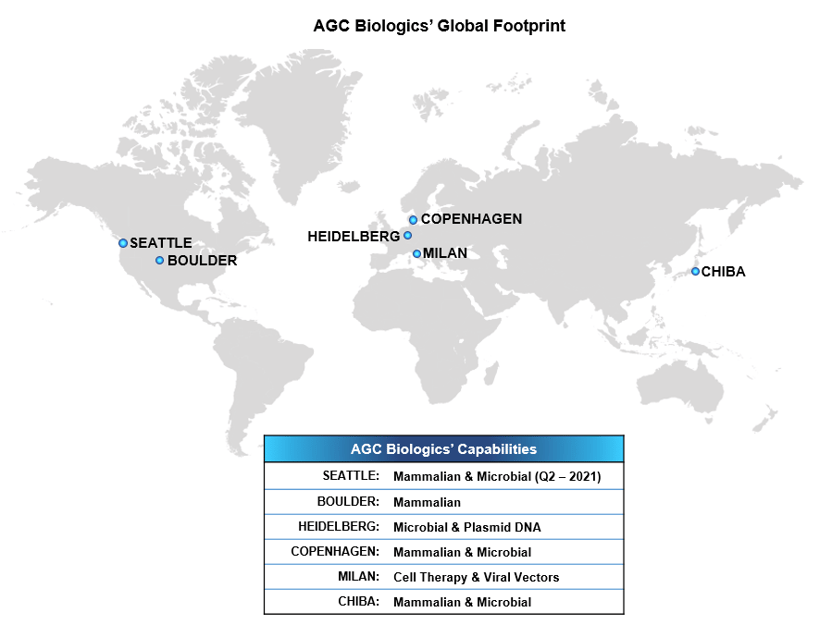
“We are continuously looking for ways to expand and extend our capabilities and capacities for our current and future customers,” says AGC Biologics Chief Executive Officer, Patricio Massera. “This significant expansion of the capacity of our Copenhagen facility will further strengthen our ability to meet the needs of our customers across our global network.”
“Building on our leadership in single use manufacturing, we are now establishing a new state of the art facility with a flexible and efficient set-up for biologic drug substance manufacturing. It will be a great facility and we are looking forward to making it available for existing and new Customers,” says Chief Technical Officer, Kasper Møller.
About AGC Biologics:
AGC Biologics is a leading global biopharmaceutical Contract Development and Manufacturing Organization (CDMO) with a strong commitment to deliver the highest standard of service as we work side-by-side with our clients and partners, every step of the way. We provide world-class development and manufacture of mammalian and microbial-based therapeutic proteins, plasmid DNA (pDNA), viral vectors and genetically engineered cells. Our global network spans the U.S., Europe and Asia, with cGMP-compliant facilities in Seattle, Washington; Boulder, Colorado; Copenhagen, Denmark; Heidelberg, Germany; Milan, Italy; and Chiba, Japan and we currently employ more than 1,600 employees worldwide. Our commitment to continuous innovation fosters the technical creativity to solve our clients’ most complex challenges, including specialization in fast-track projects and rare diseases. AGC Biologics is the partner of choice. To learn more, visit www.agcbio.com.

.jpg)

