Technology Transfer
Our tech transfer services support both large pharma and small developers
Professional tech transfer to put your mind at ease
When working with a CDMO, there are stages to tech transfer designed to speed up the process while at the same time mitigating the risks of making decisions before a complete process fit is finalized.
AGC Biologics can support your technology transfer and is here to partner with you at any of the critical stages in your product's lifecycle.
Whether you are working to scale up a new product or simply need to transfer a project to one of our seven global sites for more manufacturing capacity, we can assist you.
Speak with a rep to learn moreUnderstanding the keys to successful manufacturing process transfer
Learn how to speed-up the technology transfer process while mitigating possible delays and unexpected costs.
DOWNLOAD NOWOur process
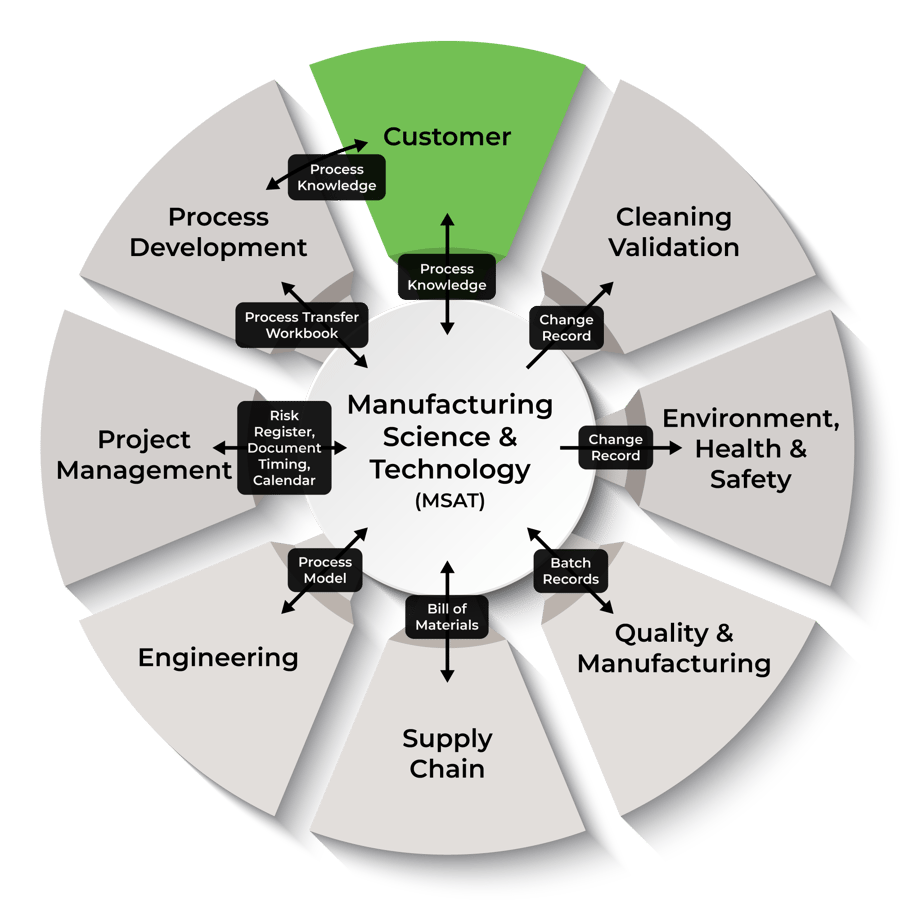
AGC Biologics follows detailed process-fitting guidelines and employs many tools to manage the complex and diverse activities associated with a technology transfer.
Our engineers and scientists help make the tech transfer process as simple, seamless, and consistent as possible. Further, we employ a flexible approach to ensure there is confidence in an efficient and successful transfer process.
Our teams of highly qualified experts have the skills and experience you can trust, and at AGC Biologics, we also provide them with additive training, development, and support. This all helps ensure you have exactly what you need throughout the entire project.
Whether you are in the development or clinical phase or need additional capacity for your commercial product, AGC Biologics is here to support you at any stage with our tech transfer services.
Interested in learning more?
Contact our teamFAQs
-
Does AGC Biologics support technology transfer?
Yes, we have specialized teams and protocols to facilitate seamless technology transfers of existing processes into our global network. We ensure process validation, gap analysis, and regulatory compliance at every stage to maintain product quality during the transfer.
-
How does AGC Biologics handle global tech transfers?
AGC Biologics utilizes a standardized global tech transfer process to ensure consistency and efficiency when moving projects between our sites or from external partners. Our global network approach allows customers to leverage capacity across multiple sites (e.g., Seattle, Copenhagen, Chiba) and a network of scientific experts, with harmonized quality systems.
-
How is digitalization transforming technology transfer at AGC Biologics?
We are actively focused on exploring and implementing digital solutions, such as electronic batch records (eBR), to streamline operations and enhance efficiency during technology transfer. These innovations allow us to maintain strategic agility and balance speed with quality, redefining the future of process development for our partners.

