Cell Therapy
Manufacturing
We offer end-to-end services, including cell culture and cell modification in process development and cGMP environments
End-to-End Cell Therapy Development and Manufacturing
We develop and manufacture diverse cell therapies, using numerous technologies, ranging from closed to open systems at different scales depending on your specific needs.
Autologous and Allogeneic Methods
Using the latest autologous and allogeneic methods, we help your products meet the exact specifications for patients in a clinical or commercial setting. We also have flexible and cost-effective services for process development and pre-clinical needs.

Cell Therapy Global Network
Our teams of experts provide the quality systems background and regulatory experience to help you navigate the important cell therapy product milestones, at any stage of the journey.

Pre-Clinical Through Commercial
We offer industry-leading expertise in process development and internalizing client cell therapy processes, including customization, development, qualification and validation activities for manufacturing and analytics. AGC Biologics’ services support every stage of the product journey, from pre-clinical through commercialization.
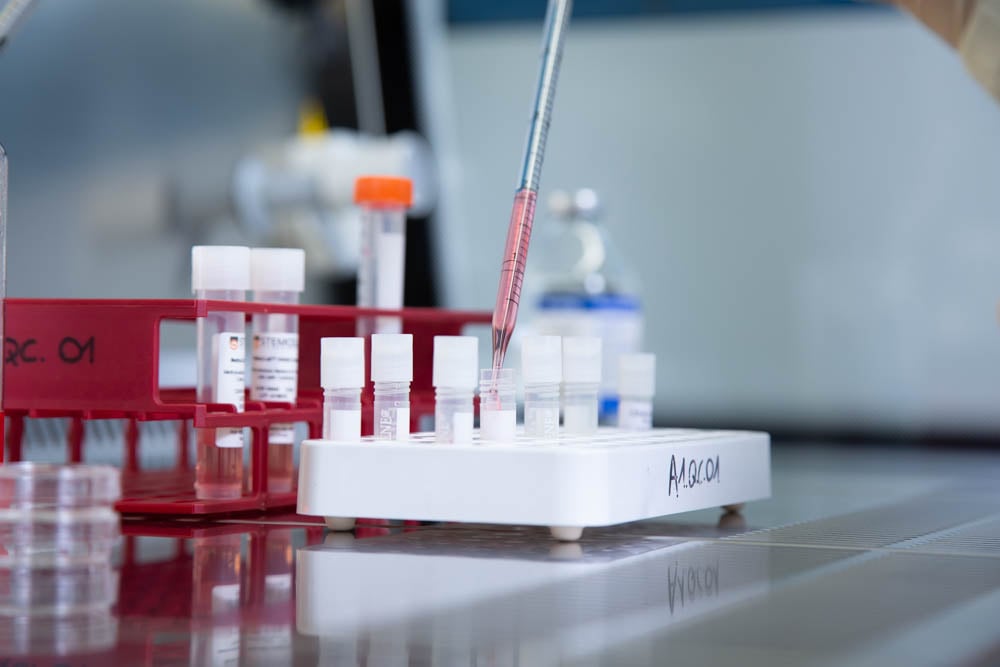
Best-in-Class Cell Therapy CDMO services
Offering top-tier cell therapy CDMO services, we excel in every stage of the life cycle, providing comprehensive support from initial development to clinical and commercial manufacturing. We specialize in cell culture and modification. Our capabilities encompass the creation of engineered cells at various scales, backed by analytical method development and qualification.
With a repertoire of over 160 in-house analytical tests thoughtfully designed for clinical and commercial supply, we stand as a comprehensive solution for cell therapy CDMO services.
Have a project in mind?
Cell Therapy Manufacturing Track Record
+0
Cell and gene therapy treated patients
0
Commercial cell therapy approvals
+0
Cell and gene therapy trials supplied in EU and US
Listen to Expert Interviews from Our Cell & Gene Center of Excellence in Milan






Click and drag video thumbnails to see more content.
Outsourcing strategies for allogeneic cell therapy
Learn about strategies for speeding up and optimizing the manufacturing of allogeneic cell therapies.
Creating cost-effective cell therapy programs with NK Cells
As this cell type continues to grow in popularity there are several strategies to consider for building a treatment program.
DownloadFill out the form to access "Opportunities to Accelerate Allogeneic Cell Therapy"
Fill out the form to access "The promise of NK cells and 3 strategies for creating a cost-effective program"
Full Lifecycle Cell Therapy Services

Cell Media
Strategic partnership with exosome cell manufacturing company RoosterBio to provide the best cell media possible.
We work with virtually any cell type, including Hematopoietic stem cells (HSCs), Human Mesenchymal Stem Cells (hMSCs), T Cells, NK Cells, and more.
95% of analytical tests performed in-house, reducing overall turnaround time and shortening your project timeline.
FAQs
-
What cell therapy services does AGC Biologics offer?
We offer services from process development through commercial manufacturing for both autologous and allogeneic cell therapies, and can support both in-vivo and ex-vivo therapy types. Our expertise covers a wide range of cell types, including CAR-T, hematopoietic stem cells (HSC), mesenchymal stem cells (MSC), induced pluripotent stem cells (iPSC), and more, all supported by our global network of flexible cGMP suites.
-
Can you support feasibility studies for cell & gene therapies?
Yes, we offer our CMC Jumpstart™ program, a rapid feasibility study package designed to assess the manufacturability of your cell or gene therapy candidate early in development. This program helps de-risk the path to GMP manufacturing. By offering a firsthand demonstration of AGC Biologics’ expertise, companies can assess how well we can meet their specific project requirements before committing to larger-scale work. Once the GOI is provided, AGC Biologics performs a representative scale-down run that provides essential Chemistry, Manufacturing, and Controls (CMC) data for a company’s top candidate.
-
What is your experience in cell & gene therapy manufacturing?
AGC Biologics has over 30 years of experience in cell and gene therapy. We have an impressive track record of commercial approvals for both viral vectors and cell therapies, making us one of the most accomplished CGT CDMOs in the industry. Additionally, we have manufactured hundreds of GMP batches for partners around the globe.








